Refractory Hypothyroidism in Pediatrics: Insights from Levothyroxine Absorption Test
Gaia Vincenzi1, Luisa Del Giacco2, Adele Matilde Tura2,Marco Abbate1, Giulia Tarantola2, Ilenia Teresa Petralia2,Cristina Santagiuliana2,Sara Zanelli2 and Maria Cristina Vigone1*
1Department of Pediatrics, IRCCS Ospedale San Raffaele, Milan, Italy.
2Department of Pediatrics, Vita-Salute San Raffaele University, IRCCS Ospedale San Raffaele, Milan, Italy
*Corresponding author
Vigone Maria Cristina , Department of Pediatrics, IRCCS Ospedale San Raffaele, Milan, Italy
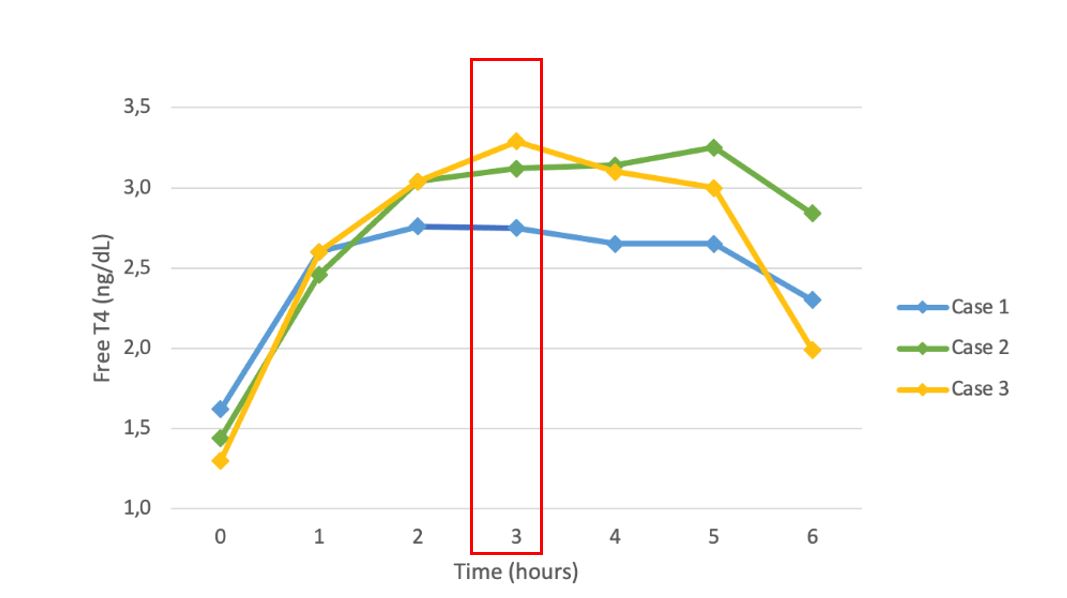
Figure 1: Comparison between the curves resulting from the levothyroxine absorption test (LT4AT) conducted in the three patients described.
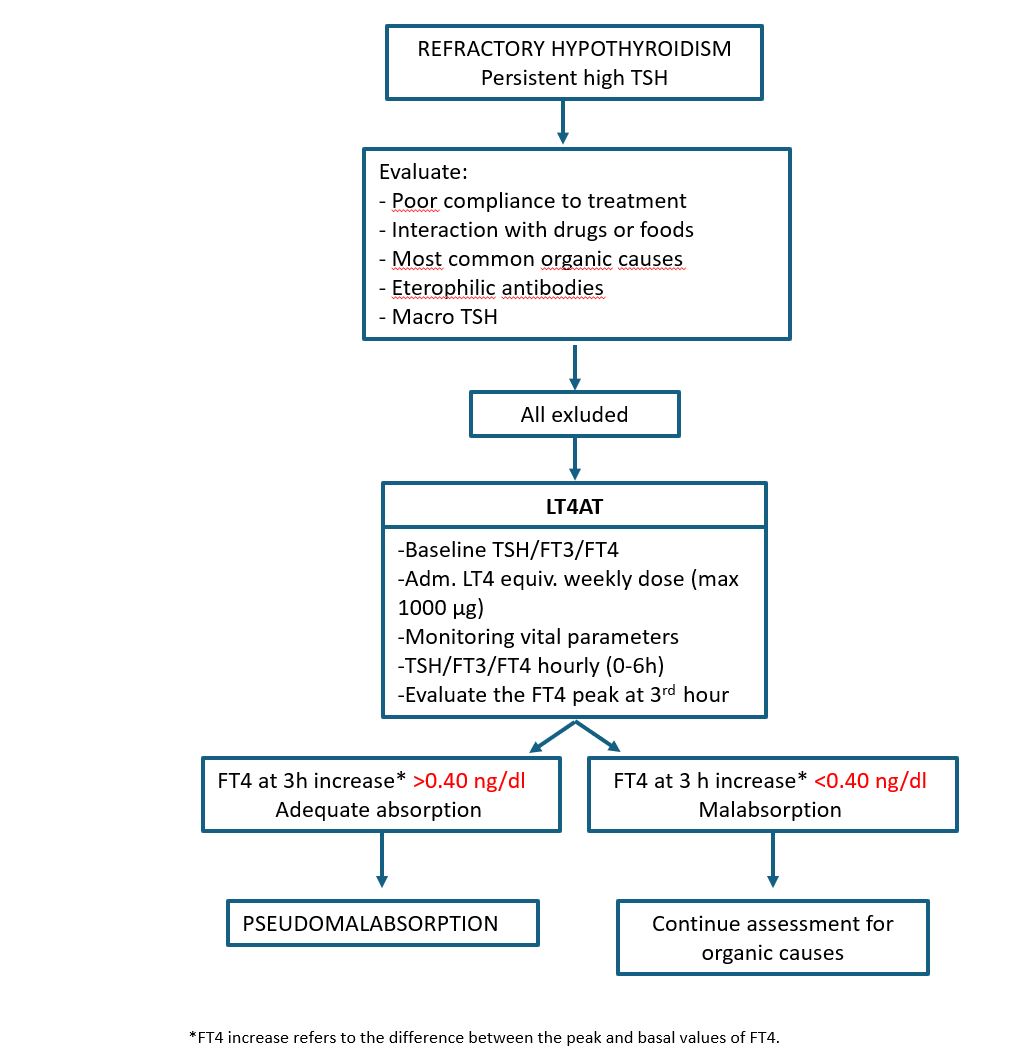
Figure 2: Diagnostic-therapeutic algorithm for refractory hypothyroidism proposed. VP: vital parameters. Adm. LT4 equiv.: administration of LT4 equivalent to the weekly dose.
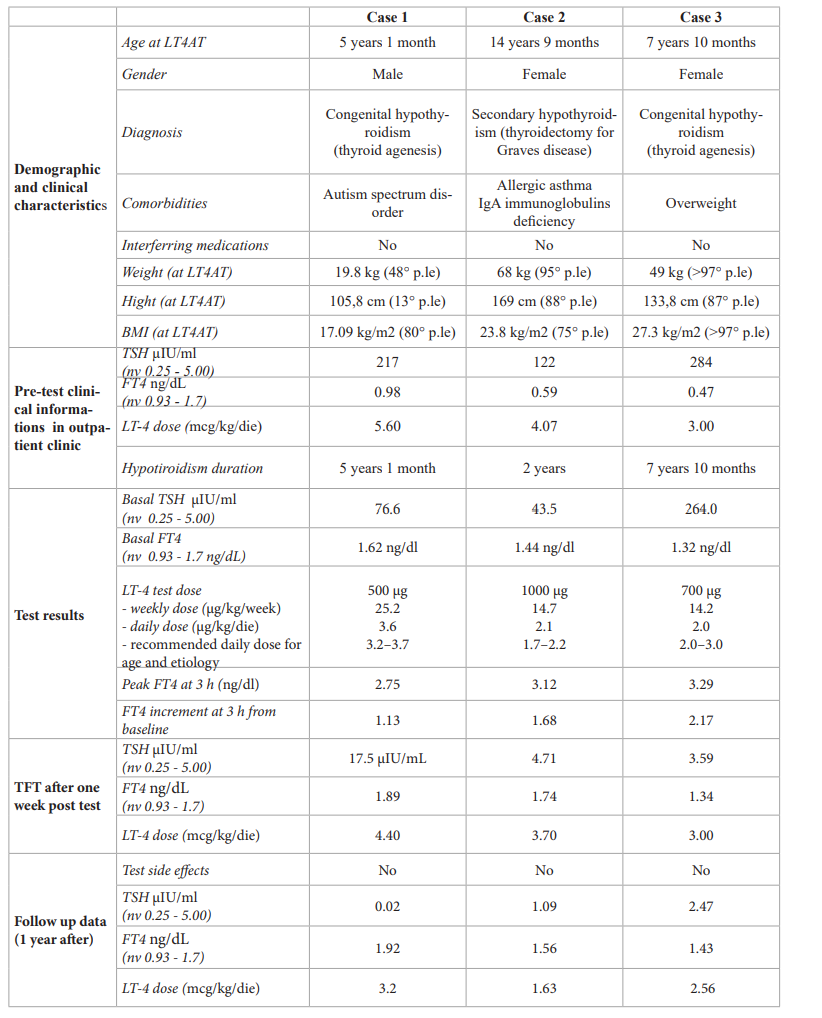
Table 1: Patients characteristics and thyroid function values before, during, and after the Levothyroxine absorption test (LT4AT). TFT: Thyroid function test; FT4: free thyroxine; FT3: free tri-iodothyronine; TSH: thyroid stimulating hormone; LT4: levothyroxine; BMI: body mass index
- Van Trotsenburg P, Stoupa A, Léger J, Rohrer T, Peters C, et al. (2021) Congenital Hypothyroidism: A 2020-2021 Consensus Guidelines Update-An ENDO-European Reference Network Initiative Endorsed by the European Society for Pediatric Endocrinology and the European Society for Endocrinology. Thyroid. 31(3): 387-419.
- Caron P, Grunenwald S, Persani L, Borson-Chazot F, Leroy R, et al. (2022) Factors influencing the levothyroxine dose in the hormone replacement therapy of primary hypothyroidism in adults. Rev Endocr Metab Disord 23(3):463-483.
- Gonzales KM, Stan MN, Morris JC 3rd, Bernet V, Castro MR (2019) The Levothyroxine Absorption Test: A Four-Year Experience (2015-2018) at The Mayo Clinic. Thyroid 29(12): 1734-1742.
- Quiroz-Aldave JE, Concepción-Zavaleta MJ, Durand-Vásquez MDC, Concepción-Urteaga LA, Gamarra-Osorio ER, et al. (2023) Refractory Hypothyroidism: Unraveling the Complexities of Diagnosis and Management. Endocr Pract 29(12):1007-1016.
- Caron P, Declèves X (2023) The Use of Levothyroxine Absorption Tests in Clinical Practice. J Clin Endocrinol Metab 108(8): 1875-1888.
- Ghosh S, Pramanik S, Biswas K, Bhattacharjee K, Sarkar R, et al. (2020) Levothyroxine Absorption Test to Differentiate Pseudomalabsorption from True Malabsorption. Eur Thyroid J 9(1):19-24.
- Lipp HP (2021) Administration and Pharmacokinetics of Levothyroxine. 2021 Mar 12. In: Kahaly GJ, editor. 70 Years of Levothyroxine.
- Yildirim Simsir I, Soyaltin UE, Ozgen AG (2019) Levothyroxine absorption test results in patients with TSH elevation resistant to treatment. Endocrine 64(1): 118-121.
- Delvecchio M, Salerno M, Vigone MC, Wasniewska M, Popolo PP, et al. (2015) Levothyroxine requirement in congenital hypothyroidism: a 12-year longitudinal study. Endocrine 50(3): 674-680.
- Virili C, Antonelli A, Santaguida MG, Benvenga S, Centanni M (2019) Gastrointestinal Malabsorption of Thyroxine. Endocr Rev 40(1): 118-136.
- Kubota S, Fukata S, Matsuzuka F, Kuma K, Miyauchi A (2003) Successful management of a patient with pseudomalabsorption of levothyroxine. Int J Psychiatry Med 33: 183-188.
- Jauk B, Mikosch P, Gallowitsch HJ (2000) Unusual malabsorption of levothyroxine. Thyroid 10(1): 93-95.
- Santos Monteiro S, Santos TS, Lopes AM, Oliveira JC, Freitas C, et al. (2022) Levothyroxine malabsorption or pseudomalab- sorption? A question in the management of refractory hypothyroid- ism. Endocr Connect 11(12): e220355.
- Soares RMV, de Figueiredo RM, Melo Dantas MN, Solano Brito MV, Pires Sousa AG, et al. (2016) Rapid Levothyroxine (Lt4) Absorption Test for Diagnosis of Lt4 Pseudomalabsorption: Case Report and Proposal of a Cutoff Point. J Endocrinol Diabetes Obes 4(1): 1083.
- Lips DJ, van Reisen MT, Voigt V, Venekamp W (2004) Diagnosis and treatment of levothyroxine pseudomalabsorption. Neth J Med 62(4):114-118.
- Barbar B, Hossen S, Quinton R, James A, Perros P, et al. (2023) Levothyroxine absorption test: A therapeutic strategy for improving medication adherence. Clin Endocrinol (Oxf) 98(5): 741-742.
- Centanni M, Benvenga S, Sachmechi I (2017) Diagnosis and management of treatment-refractory hypothyroidism: an expert consensus report. J Endocrinol Invest 40(12): 1289-1301.
- Sun GE, Pantalone KM, Faiman C, Gupta M, Olansky L, et al. (2014) The clinical utility of free thyroxine in oral levothyroxine absorption testing. Endocr Pract 20(9):925-929.